 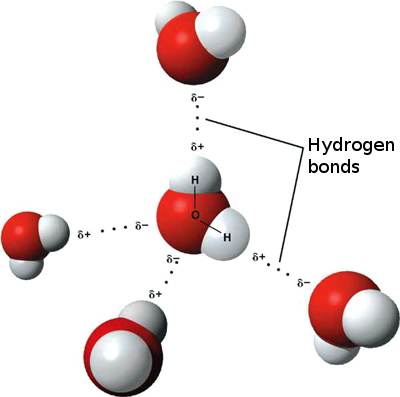
This allows direct detection and characterizations of hydrogen bonds involving Lys NH 3 + by NMR. Owing to the very slow 15N transverse relaxation of NH 3 + groups, which permits the use of a long period for J-modulation, hydrogen-bond scalar coupling constants in the range of 0.1–1.0 Hz can readily be measured for Lys side chains of midsize (~ 30 kDa) systems studied by NMR. An analysis of hydrogen bonding can give important clues in determining the correct orientation of an ambiguous side chain ( Fig. In a similar manner, the orientations of histidines, threonine, glutamine, and asparagine are ambiguous in protein maps where the slight density difference between a carbon, oxygen, or nitrogen atom cannot be safely distinguished. Histidines can have various protonation states, and an analysis of the hydrogen bonds can allow a determination of the most likely protonation state according to whether hydrogen is bonded to a donor or to a acceptor. Water is especially facile at hydrogen bonding because it is both an acceptor and a donor. Hydrogen bonds often occur in networks-frequently with water mediating. The closer the hydrogen bond is to correct geometry, the stronger the bond. 43 The angle the bond forms is also important in determining the strength of the hydrogen bond. This distance is generally considered to be from 2.7 to 3.3 Å, with 3.0 Å being the most common value for protein and water hydrogen bonds. If a proper hydrogen bond acceptor–donor pair is within the correct distance, the bond is taken to be a hydrogen bond. Hydrogen bonds are found in protein crystallography indirectly. In the latter half of the century, strong hydrogen bonds were discovered in several classes of organic molecules in crystals and aprotic liquid phases and characterized by crystallographic, spectroscopic, and chemical methods. Such strong hydrogen bonds were thought to be limited to crystalline states. Very few strong hydrogen bonds had been documented before the mid-twentieth century, principally the very strong hydrogen bond in hydrogen difluoride − (40 kcal mol −1). Unlike covalent bonds, which vary in strength within a factor of ~4 (30–120 kcal mol −1), hydrogen bonds are much less constrained in their geometry and physical properties, and they vary in strength by a factor of at least 20-fold (2–40 kcal mol −1). By mid-twentieth century, the weak conventional hydrogen bonds were reasonably well understood and widely accepted. Hydrogen bonds were controversial throughout the twentieth century. Frey, in Encyclopedia of Biological Chemistry (Second Edition), 2013 The Nature of Hydrogen Bonds 68 According to this analysis, the hydrogen bond is destabilizing, no matter whether it is exposed (Δ G 1=+13 kJ mol −1) or buried (Δ G 3=+16 kJ mol −1). 68 It follows that transfer of hydrogen-bonded groups from water to CCl 4 is also unfavorable: Δ G 2=+3 kJ mol −1. 67 On the other hand, because of the dehydration penalty, transfer of NMA from water to CCl 4 is highly unfavorable, with Δ G 4=+26 kJ mol −1. It was observed that the formation of the NMA dimer is disfavored in water (Δ G 1=+13 kJ mol −1) but favored in nonaqueous solvent CCl 4 (Δ G 5=−10 kJ mol −1). For example, formation of the N-methylacetamide (NMA) dimer is a popular model for peptide hydrogen bond. 3,6 In his influential review, Dill elaborated the thermodynamics cycle of hydrogen bond formation as in Figure 6. It was believed that the protein-water hydrogen bond more or less cancels out the stabilization by the protein-protein hydrogen bond so that the contribution of the hydrogen bond to protein stability was expected to be small. 65,66 The main concern is that in order to form protein-protein hydrogen bonds, the polar groups have to break their hydrogen bonds with water. There is no question that the formation of the hydrogen bond itself is favorable – the energy of an amide-amide hydrogen bond in vacuum, estimated by quantum chemistry, is −25 kJ mol −1. Despite the ubiquitous nature of the hydrogen bond in proteins, whether it stabilizes proteins is controversial. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
